Autores
dos Santos Filho, J.M. (UFPE) ; Pinheiro, S.M. (UFPB) ; Albuquerque Barros, R.A. (UFPE)
Resumo
Amino acids constitute the building blocks of proteins, hormones, and toxins,
aside from being the formation units of neurotransmitters and nucleic bases,
e.g., adenine and guanine. Despite their importance, the main core
of the essential α-amino acids includes only 20 structurally simple molecules.
These remarkable substances are easily recognized, absorbed, and metabolized by
living beings, which base their complexity upon them. Even structurally modified
amino acids play an important role in the metabolic pathways, since their
structural backbone remains recognizable by the biomolecules ruling the cells.
Therefore, it is not surprising to suppose that modifications in the amino
acid structure aiming to incorporate recognized bioactive moieties can lead to
new drugs.
Palavras chaves
DNP-glycine; Thiosemicarbazides; Antitumor activity
Introdução
Amino acids (AA) are the building blocks of all proteins found in living beings
and, therefore, their chemical structures are easily recognized by the
biomolecules regulating cell processes. Beside this primary role, AA are also
the leitmotiv for the biosynthesis of several bioactive small molecules,
essential for life maintenance and regulation. For example, tyrosine is
converted to catecholamines, a group of three substances comprising
dopamine, norepinephrine, and epinephrine, which exert effects as either a
neurotransmitter or as a hormone in numerous parts of the body. Tryptophan is
the precursor for the synthesis of serotonin and melatonin, while histidine
leads to histamine, all potent neurotransmitters. Other AA are of similar
importance for the biosynthesis of regulatory biomolecules [KAMBLE et al,
2021]. Due to their biological importance, AA are frequently hybridized with
several privileged structures and/or well-known pharmacophoric groups in an
attempt to disclose new bioactive substances [ARUNADEVI and RAMAN,
2020]. In fact, promising investigations in this field can
be easily found in the literature, comprising several biological responses,
especially antitumor activities [CASTRO et al, 2020]. One of the most
ancient AA modifications is of central importance for the development of the
synthesis and structural
elucidation of peptides, consisting of the attachment of the amino group to the
2,4-dinitrophenyl (DNP) moiety via a SNAr reaction, leading to N
α-2,4-dinitrophenyl amino acids (DNP-AA) [ SANGER, 1945]. The
nitroaromatic portion present in many compounds with great therapeutic
significance is essential to the biological activity so that its removal leads
to the potency’s loss or even the complete lack of pharmacological response
[NEPALI et al, 2019]. Despite the DNP-AA having been known for a long
time in the literature, no investigation of their biological properties has been
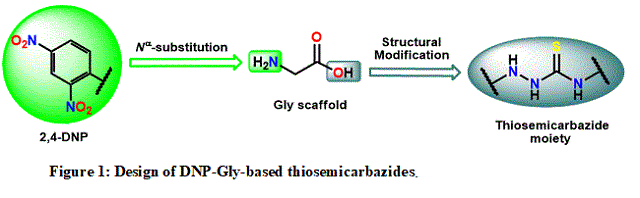
carried out until this point. Dinitrophenyl glycine (DNP-Gly) derivatives
synthesis is a relatively simple process, whose isolation and purification are
quite easy, allowing us to obtain them as pure samples. The chosen strategy for
this work has envisaged the design of derivatives bearing the thiosemicarbazide
moiety due to its importance in medicinal chemistry [ACHARYA et al,
2021], particularly as antitumor bioactive molecules [SALIB et al, 2016].
A series of DNP-Gly thiosemicarbazide derivatives were designed, synthesized,
and characterized, being expected to exhibit an antitumoral response. The
structural Gly modification design is depicted in Figure 1.
Material e métodos
Reactions’ progress was monitored by thin-layer chromatography (TLC), performed
onto glass-backed plates of silica gel 60 F254 with gypsum from Merck, and all
compounds were detected by ultraviolet light (254 nm). Melting points were
determined with a capillary apparatus and are uncorrected. Nuclear Magnetic
Resonance (NMR) spectra were recorded at 400 MHz for hydrogen (1H)
and
100 MHz for carbon-13 (13C). Analyses were determined at 25 °C in
DMSO-d6 with chemical shift values (δ) in parts per million (ppm)
and coupling constants (J) in Hertz (Hz). 1H
NMR and 13C NMR assignments were assisted by 2D experiments. Infrared
(IR) spectra were recorded on a FTIR spectrometer from Bruker with the samples
being analyzed as KBr pellets. Elemental analyses were performed in a Perkin
Elmer elemental analyzer.
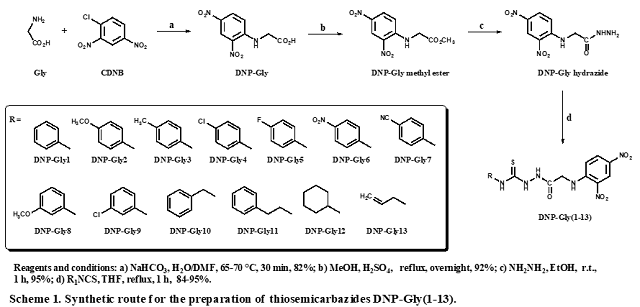
The synthesis of the substituted thiosemicarbazides DNP-Gly(1-13), depicted in
Sheme 1, was based on the structural modifications of the
commercially available amino acid glycine (Gly) as starting material. A
nucleophilic aromatic substitution (SNAr) reaction between Gly and 1-
chloro-2,4-dinitrobenzene (CDNB) was carried out, leading to the N
α-(2,4-dinitrophenyl)-glycine (DNP-Gly) after careful isolation under
appropriate conditions in good yield and high purity of crude product, avoiding
tedious
work-up. Afterward, a simple Fisher esterification of compound DNP-Gly in the
presence of methanol and mineral acid as catalyst has resulted in the
corresponding DNP-Gly methyl ester. The key DNP-Gly hydrazide was easily
prepared
in presence of hydrazine hydrate and short reaction time.
The DNP-Gly hydrazide has undergone a smooth addition reaction with suitable
isothiocyanates in THF under mild conditions, leading to a set of structures
encompassing the thiosemicarbazide series DNP-Gly(1-13). Once all experiments
were concluded, the pure products were confirmed by means of IV and elemental
analysis, as well as by NMR spectroscopy. Exact signal assignments for the new
molecules were assisted by 2D NMR experiments prior to submission to the
biological evaluation.
Resultado e discussão
The planned synthetic route of Scheme 1 was successfully carried out in 4
steps. The SNAr reaction between Gly and CDNB has occurred smoothly,
leading to the DNP-Gly with 82% yield. The Fischer
esterification has introduced the next modification, also with an excellent
outcome after a simple work-up, giving the DNP-Gly methyl ester with 92% yield.
The key intermediate DNP-Gly hydrazide has been also obtained as a yellow solid
and 95% yield of the crude product. The thiosemicarbazides DNP-Gly(1-13) has
been
readily
prepared under mild conditions by reacting DNP-Gly hydrazide with appropriate
isothiocyanates, in order to introduce a diversity of substituents in the
series. Such substituents can help the biological evaluation and the
establishment of the structure-activity relationship (SAR) arising from the
biological results. After isolation of pure products DNP-Gly(1-13) some
physicochemical data can be described.
DNP-Gly1: Yield 95%; Mp 212.5-214.2 °C; Rf 0.33 (AcOEt); IR
(KBr, cm-1): 3351, 3175 (NH), 3088 (Ar CH ), 2986, 2934 (Aliphatic
CH), 1702 (C=O), 1620 (C=C); DNP-Gly2: Yield 90%; Mp 197.7-199.0 °C;
Rf 0.58 (AcOEt); IR (KBr, cm-1): 3348, 3181 (NH), 3109 (Ar
CH), 3000 (Aliphatic CH), 1705 (C=O), 1622 (C=C); DNP-Gly3: Yield 92%; Mp
202.1-203.0 °C; Rf 0.49 (AcOEt); IR (KBr, cm-1): 3338,
3187 (NH), 3002 (Ar CH ), 2926 (Aliphatic CH), 1706 (C=O), 1621 (C=C); DNP-
Gly4: Yield 86%; Mp 203.3-204.8 °C; Rf 0.82 (AcOEt); IR (KBr,
cm-1): 3349, 3211, 3155 (NH), 3104 (Ar CH), 2979 (Aliphatic CH), 1704
(C=O), 1621 (C=C); DNP-Gly5: Yield 91%; Mp 213.9-215.0 °C; Rf
0.51 (THF/Hexanes 7:3); IR (KBr, cm-1): 3351, 3196 (NH), 3091 (Ar
CH), 2982 (Aliphatic CH), 1703 (C=O), 1620 (C=C); DNP-Gly6: Yield 90%; Mp
205.3-205.5 °C; Rf 0.32
(AcOEt/MeOH 7:3); IR (KBr, cm-1): 3327, 3301 3175 (NH), 3105, 3001
(Ar CH), 1708 (C=O), 1617 (C=C); DNP-Gly7: Yield 93%; Mp 207.2-208.7 °C;
Rf 0.29 (AcOEt/MeOH 9:1); IR (KBr, cm-1): 3451, 3327, 3150
(NH),
3106, 3042 (Ar CH ), 2957, 2901 (Aliphatic CH), 2229 (C≡N), 1704 (C=O), 1621
(C=C); DNP-Gly8: Yield 93%; Mp 173.7-174.9°C; Rf 0.31
(AcOEt/Hexanes
7:3); IR (KBr, cm-1): 3341, 3184 (NH), 3005 (Ar CH ), 2976 (Aliphatic
CH), 1705 (C=O), 1621 (C=C); DNP-Gly9: Yield 92%; Mp 203.8-205.2°C;
Rf
0.15 (THF/Hexanes 7:3); IR (KBr, cm-1): 3345, 3208 (NH), 3093 (Ar CH
), 2984 (Aliphatic CH), 1703 (C=O), 1620 (C=C); DNP-Gly10: Yield 88%; Mp
206.9-207.5 °C; Rf 0.51 (AcOEt); IR (KBr, cm-1): 3315,
3142 (NH),
3029 (Ar CH
), 2939 (Aliphatic CH), 1718 (C=O), 1623 (C=C); DNP-Gly11: Yield 92%; Mp
207.6-208.8 °C; Rf 0.46 (AcOEt); IR (KBr, cm-1): 3351,
3265, 3150
(NH), 3106, 3024 (Ar CH), 2978, 2948 (Aliphatic CH), 1695 (C=O), 1623 (C=C);
DNP-Gly12: Yield 91%; Mp 205.5-206.6 °C; Rf 0.31 (AcOEt); IR
(KBr,
cm-1): 3333, 3186, (NH), 3107 (Ar CH), 2932, 2854 (Aliphatic CH),
1703 (C=O), 1621 (C=C); DNP-Gly13: Yield 84%; Mp 194.4-195.5 °C;
Rf 0.65
(AcOEt); IR (KBr, cm-1): 3322, 3172 (NH), 2980, 2853 (Aliphatic
CH), 1719 (C=O), 1623 (C=C).
Conclusões
Herein a successful strategy was applied in the synthesis of a series of 13 N-
(2,4-dinitrophenyl)-glycine thiosemicarbazides, designed as potential antitumor
molecules. The series DNP-Gly(1-13) was fully characterized by several
spectroscopic techniques after purification. The incorporation of the glycine
framework in the series
of DNP-Gly thiosemicarcarbazides represents an outstanding strategy for the search
of new
bioactive compounds, opening the possibility of discovering new lead molecules
with innovative structural features.
Agradecimentos
The authors are grateful to Mrs. Eliete de Fátima V. B. N. da Silva and the
Analytical Centre of Fundamental Chemistry Department, Universidade Federal de
Pernambuco, for the NMR, IV, and elemental analysis experiments.
Referências
ACHARYA, P.T., BHAVSAR, Z.A., JETHAVA, D.J., PATEL, D.B., PATEL, H.D. A review on development of bio-active thiosemicarbazide derivatives: Recent advances, J. Mol. Struct. 1226 (2021) 129268.
ARUNADEVI, A., RAMAN, N., Biological response of Schiff base metal complexes incorporating amino acids-a short review, J. Coord. Chem. 73 (15) (2020) 2095-2116.
DE CASTRO, P.P., SIQUEIRA, R.P., CONFORTE, L., FRANCO, C.H.J., BRESSAN, G.C., AMARANTE, G.W. Cytotoxic Activity of Synthetic Chiral Amino Acid Derivatives, J. Braz. Chem. Soc. 31 (1) (2020) 193-200.
KAMBLE, C., CHAVEN, R., KAMBLE, V. A Review on Amino Acids, Res. Rev. J. Drug Des.
Discov. 8 (3) (2021) 19-27.
NEPALI, K., LEE, H.-Y., LIOU, J.-P., Nitro-Group-Containing Drugs, J. Med. Chem. 62 (2019) 2851-2893.
SALIB, S. B., KHALIL, O. M., KAMEL, M. M., EL-DASH, Y. Synthesis and Antitumor Activity of Novel Thienopyrimidine Derivatives Containing Thiosemicarbazide Moiety, OALib Journal 3 (2016), 1-7.
SANGER, F., The Free Amino Groups of Insulin, Biochem. J. 39 (1945) 507-515.
















